 Other studies of drug-eluting stent thrombosis have produced similar findings. 
In addition, narrower stents (≤2.75 mm in diameter before deployment) were more likely to be underexpanded. In a smaller study of IVUS findings from 50 patients, Taherioun and colleagues 6 noted suboptimal stent deployment in 11 patients (22%), even after adjunctive postdilatation with a noncompliant balloon stent underexpansion was noted in 7 of those 11 patients. 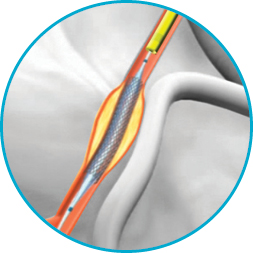
4 In a large retrospective study of post-PCI intravascular ultrasound (IVUS) findings in 7,484 patients, Cheneau and colleagues 5 identified 23 stented lesions in which subacute thrombosis occurred stents were underexpanded in 18 (78%). Thrombosis in underexpanded stents can be more severe and diffuse than in-stent restenosis and usually involves the stent's proximal part. 3, 4 Two major complications of stent underexpansion are acute or subacute stent thrombosis and in-stent restenosis. Stenting and completely dilating a complex calcified coronary artery lesion can be challenging and may result in stent underexpansion. At the patient's 3-month follow-up visit, he was doing well and had an LVEF of 0.40 to 0.45. The noncompliant balloon was then inflated at a pressure of 24 atm for 7 s, fully expanding the stent with a good final angiographic result ( Fig. Next, a new 3.5 × 12-mm NC Trek noncompliant balloon (Abbott Vascular) was placed on the Runthrough NS guidewire and advanced over the 2 BMW guidewires across the stent ( Fig. Consequently, the 6F GuideLiner catheter was retracted and exchanged for 2 Balance Middleweight (BMW) guidewires (Abbott Vascular), which were then maneuvered across the underexpanded stent. A 3.5 × 10-mm Flextome cutting balloon (Boston Scientific Corporation) was then maneuvered to the stent but could not be moved inside, despite aggressive support with the guidewire and 6F GuideLiner catheter. 1A), 70% in the ostial first diagonal branch, 30% in the proximal LCx, and 70% in the distal LCx, and a patent stent in the RCA.Ī second, unsuccessful attempt was made to fully expand the stent by inserting a 3.5-mm noncompliant balloon and inflating it at a pressure of 24 atm for 10 s ( Fig. The resulting coronary angiogram showed 30% stenosis in the distal left main coronary artery, 90% in the mid-to-distal left anterior descending coronary artery (LAD) ( Fig. Because the newly decreased LVEF was accompanied by acute decompensated heart failure, coronary angiography was performed after diuresis. Solutions, Minneapolis, Minnesota, USA) to successfully treat a case of CTO performed through. An echocardiogram revealed a left ventricular ejection fraction (LVEF) of 0.35 to 0.40. applied the Guideliner mother-and-child catheter (Vascular. A chest radiograph showed interstitial pulmonary edema. An electrocardiogram (ECG) revealed sinus rhythm, an old inferior myocardial infarction, and new anterior ST-segment depressions when compared with a baseline ECG. The Company offers catheter, hemostat and vein products to interventional cardiologists, radiologists, and vascular surgeons worldwide. Laboratory evaluations revealed an elevated brain natriuretic peptide level of 1,277 pg/mL (normal, ≤450 pg/mL) and a normal troponin I level. Vascular Solutions, a Teleflex company, operates as a medical device company that focuses on developing clinical solutions for coronary and peripheral vascular procedures. Physical examination revealed normal vital signs, jugular venous distention, and rales at the base of the lungs. His medical history included coronary artery disease, type 2 diabetes, hypertension, hyperlipidemia, smoking, and myocardial infarction treated by PCI in the left circumflex coronary artery (LCx) 10 years previously and the right coronary artery (RCA) 12 years previously. 
The terms of the settlement are confidential.A 69-year-old man presented with new-onset New York Heart Association (NYHA) class III functional status and shortness of breath of 3 weeks' duration. As part of the settlement agreement, all litigation between the two parties related to guide extension will be dismissed. Boston Scientific filed a counterclaim in July 2013, alleging that Vascular Solutions infringed a patent owned by Boston Scientific related to rapid exchange guide extension technology by manufacturing and selling its GuideLiner guide extension catheter. District Court for the District of Minnesota in May 2013, alleging that Boston Scientific infringed three patents owned by Vascular Solutions related to rapid exchange guide extension technology by manufacturing and selling its Guidezilla guide extension catheter. Virtually every substantive aspect of our GuideLiner product and patents, from the design to the dimensions to even the exact words used in the products. Vascular Solutions filed the original lawsuit in U.S. today announced that it has reached a settlement of its patent lawsuit with Boston Scientific Corporation relating to patents in the area of coronary guide extension catheters. 
MINNEAPOLIS, Minnesota - Vascular Solutions, Inc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
